Most quality teams in the UAE and MENA are stuck between enterprise platforms that take months to implement and Excel sheets that break down the moment something goes wrong. flowdit is the middle ground — digital audits, inspection checklists, nonconformance tracking, and closed-loop CAPA in one platform your team can use from day one. No IT project, no long onboarding, no six-figure price tag.




Teams using flowdit complete audits up to 30% faster – with automated scheduling, digital checklists, and instant PDF reports replacing manual paperwork.
Reduce scrap and error rates, and the associated complaints, leading to greater customer satisfaction and loyalty.
The platform's ease of use boosts employee satisfaction and productivity, resulting in lower turnover.
flowdit supports Arabic and is built for organizations across MENA, the UAE, and the Middle East, aligning with regional regulations and local operational requirements.
See how quality teams replace scattered spreadsheets and manual follow-ups with one connected system – from the first audit finding to the closed corrective action, with a full audit trail throughout.
Stop juggling spreadsheets, email threads, and disconnected tools. flowdit Audit brings quality management into one platform–from audit planning and execution to findings, CAPA, and reporting. Whether you manage process audits, product quality checks, LPA, 5S, or safety requirements, you maintain a single source of truth aligned with ISO, customer, and internal quality standards.
Designed for ISO 9001, IATF 16949, and internal quality standards
Plan, schedule, and execute quality audits from a central system
Identify quality risks early and trigger CAPA automatically
Consistent quality results across sites, plants, and shifts
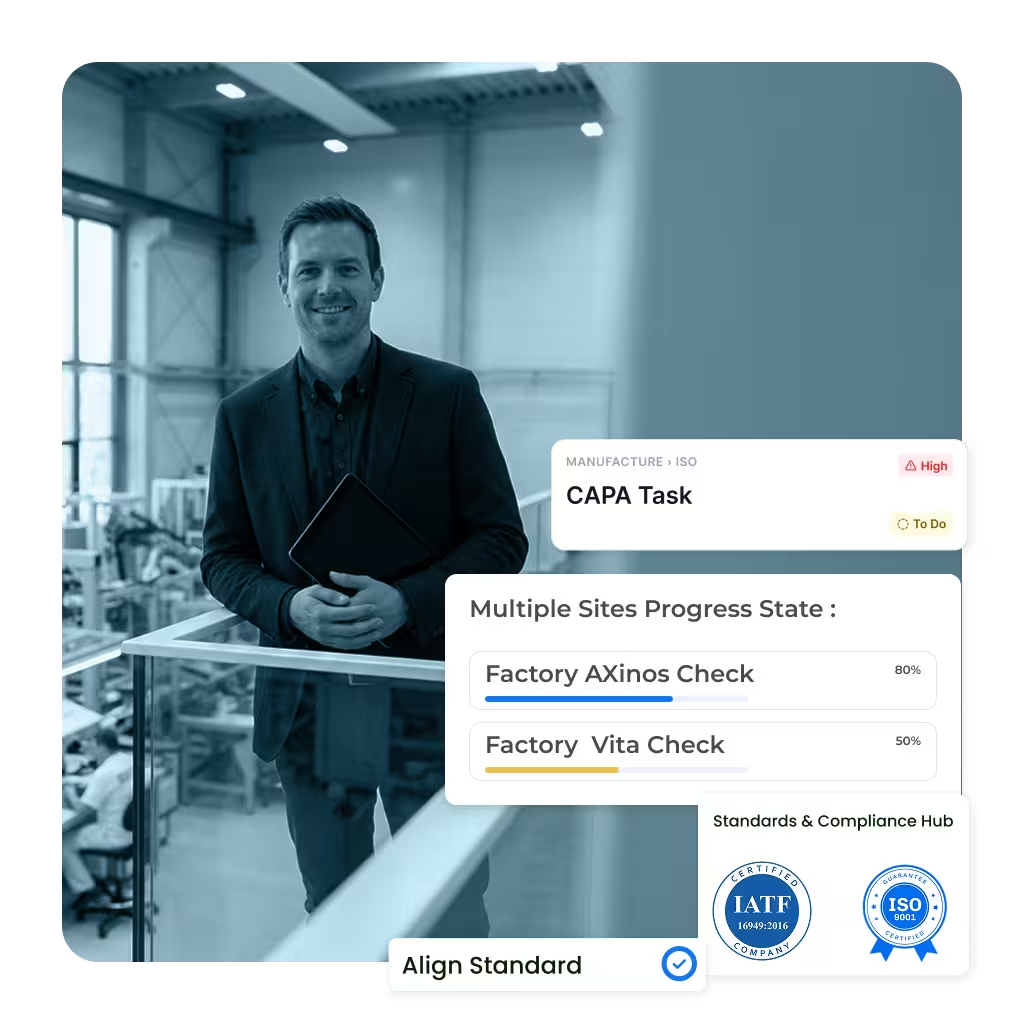
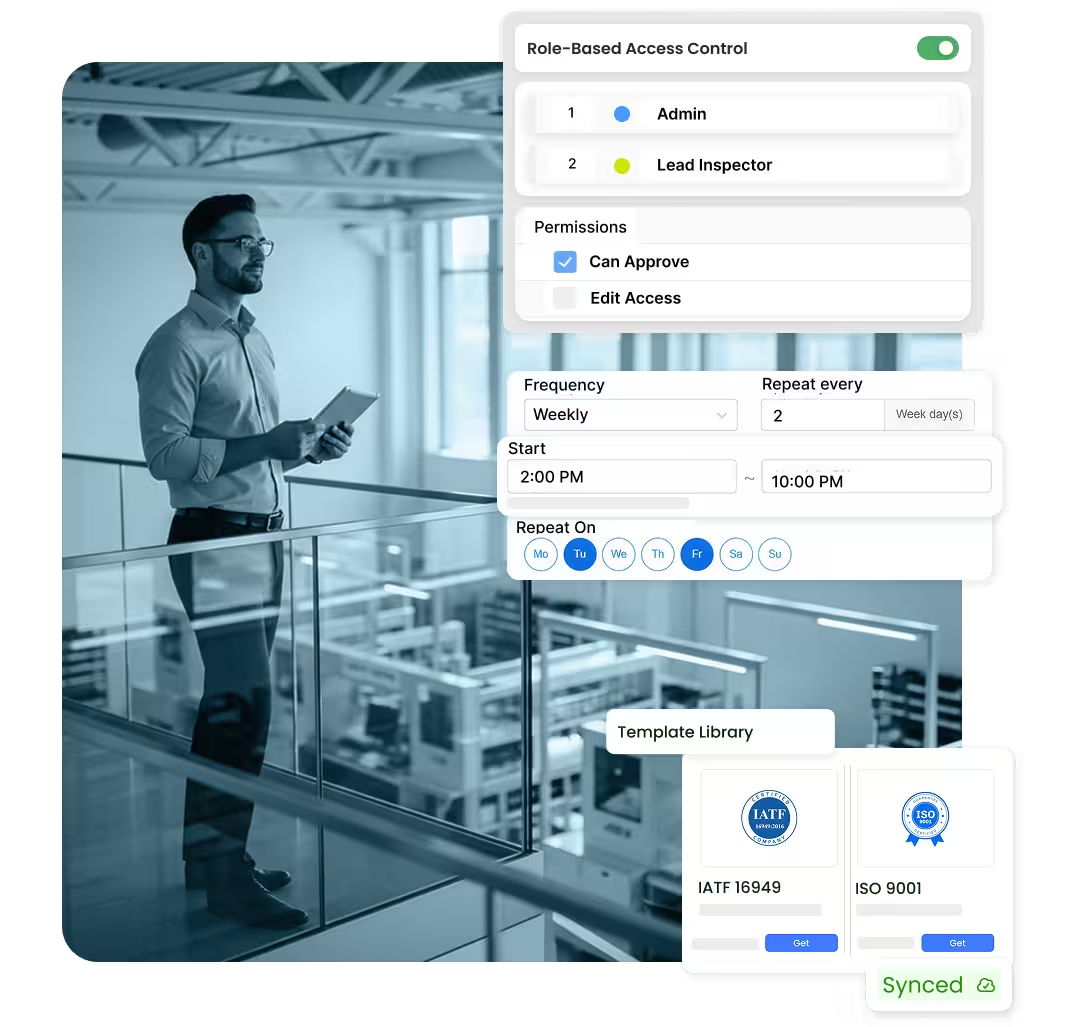
Your quality management software should support daily operations, not create additional overhead. flowdit Audit automates scheduling, permissions, reminders, and quality templates. Auditors and quality teams capture findings directly on mobile devices, add photos and notes, and track quality status in real time–without manual Excel tracking.
Role-based access for multi-site quality organizations
Standardized quality templates for recurring audits and inspections
Automated scheduling, escalation, and reminders
Desktop, tablet, and mobile quality workflows – online or offline
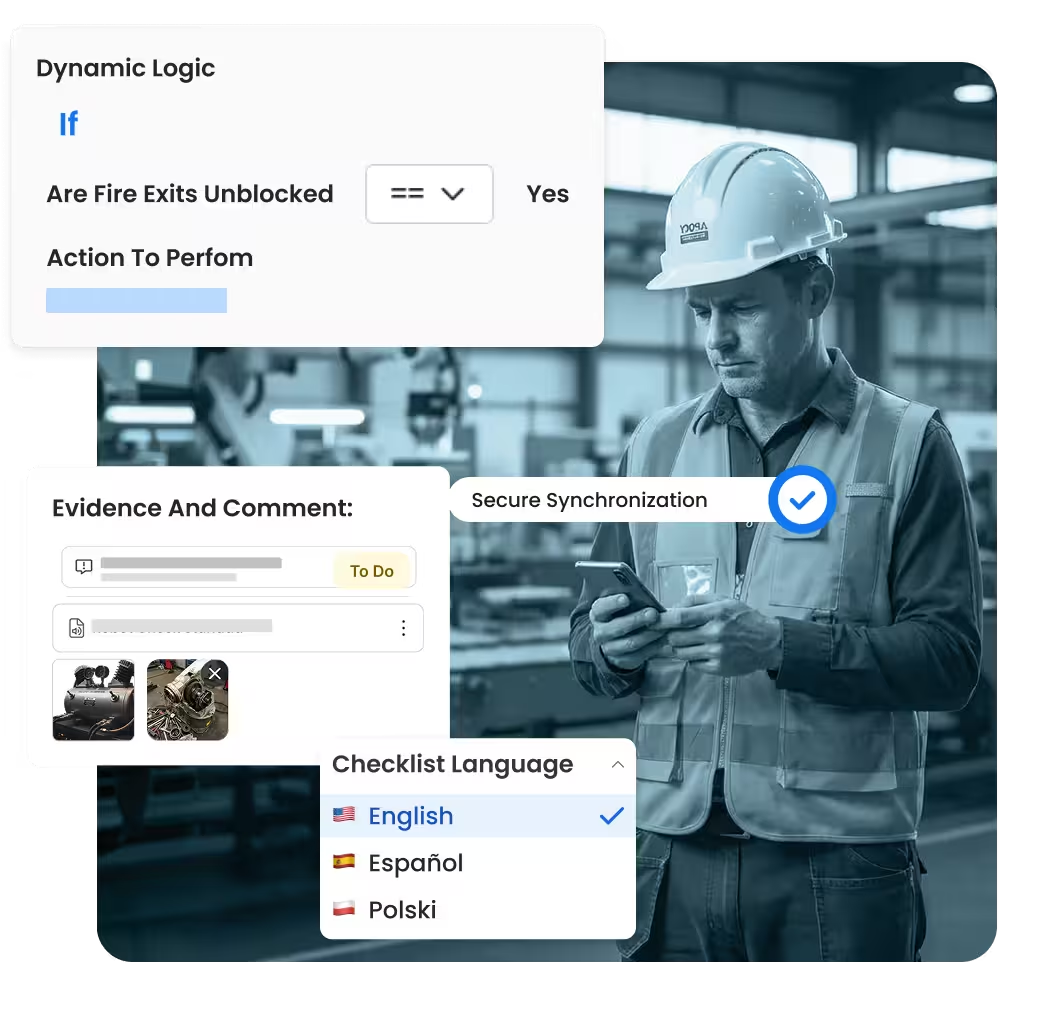
Replace static paper forms with digital quality checklists that guide teams step by step. Conditional logic, required evidence, and weighted scoring ensure quality audits and inspections are complete, comparable, and audit-ready–online or offline on the shop floor.
Conditional logic, scoring, and mandatory quality questions
Capture photos, notes, and documents at the point of inspection
Offline-first quality app with secure synchronization
Multi-language quality checks with lookups for standards and assets
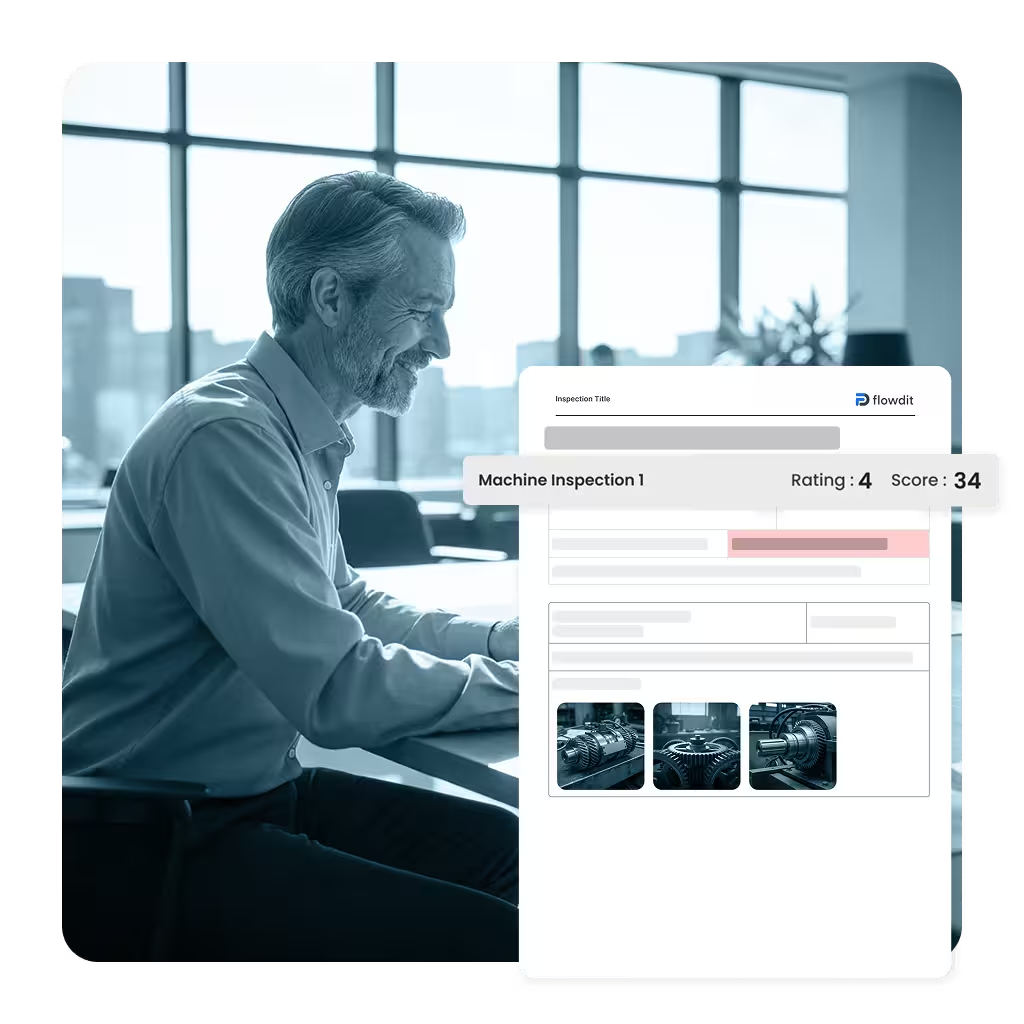
Eliminate manual reporting work. flowdit Audit applies your quality scoring models, calculates results automatically, and generates standardized, branded PDF reports. Every quality audit includes a complete audit trail with approvals and e-signatures–keeping you prepared for customers, certification bodies, and internal reviews.
Configurable scoring and grading per quality audit type
Immutable quality audit trail with approvals and e-signatures
Branded PDF quality reports aligned with corporate standards
Share quality results securely via email or links
Every quality audit and inspection generates valuable data. With quality management software from flowdit, you export structured quality data to Excel, CSV, or BI tools to analyze trends, compare sites, and identify recurring nonconformities. Drive continuous improvement using facts–not assumptions.
Export quality data to Excel, CSV, and BI tools
KPIs, trends, and heatmaps for quality transparency
Drill-down by site, process, product, or audit field
APIs to integrate with ERP, QMS, and core business systems
Quality audits only create value when corrective actions are completed. flowdit Audit links every quality finding to CAPA tasks with clear ownership, deadlines, and evidence. Track corrective actions centrally, receive automated reminders, and verify effectiveness–so quality improvements are sustained, not forgotten.
CAPA board connected directly to quality findings
Automatic reminders and escalation for overdue CAPA
Evidence upload and approval workflows
Reusable CAPA templates for recurring quality issues
Process audits, product quality checks, LPA, 5S, and safety requirements: plan, execute, and follow up using one quality management software. Secure, scalable, and aligned with your compliance and certification standards.
Online & offline ready for shop-floor audits
Templates for common internal and supplier audits
Role-based rights for multi-site organizations
Exports and APIs for BI, ERP and QMS tools
Quality management in one platform, from audits and inspections to CAPA, reporting, and analysis.
Everything you need to know about getting started with flowdit Audit.
Prefer email instead of a call? Share your audit goals and we’ll respond within one business day with concrete next steps.